We have a bipartisan Omnibus bill

From Capitol Hill, the Washington Post reports
The Senate on Tuesday took the first formal step toward advancing a bipartisan, roughly $1.7 trillion deal to fund the U.S. government, as Democrats and Republicans raced to avert a shutdown in the final days of the year.
Lawmakers voted 70-25 to begin debate on the 4,155-page measure, known in congressional parlance as an omnibus, which would fund key elements of President Biden’s economic agenda, boost defense programs, and provision an additional $44.9 billion in emergency military and economic assistance for Ukraine.
The lumbering Senate sought to move with uncharacteristic haste after congressional leaders released the full text of the bill in the early hours of the morning, capping off months of intense legislating.
Becker’s Hospital CFO Review, the American Hospital Association, and Politico Pulse offer healthcare takeaways from the omnibus. Of note, Congress laid the groundwork for a soft landing following the public health emergency by addressing Medicaid, the AMA’s concern about the impending Medicare Part B cut (“narrowing the cut to 2 percentage points in the year ahead with a scheduled cut of 3.25 percentage points in 2024″) and extending Medicare telehealth flexibilities and most other Pandemic tied flexibilities through 2024.
Govexec and Federal News Network provide omnibus insights on federal agency and employment issues. Of note, Congress implicitly gave the green light to a 4.6% raise for federal employees in 2023, broken out into a 4.1% across-the-board increase and the remainder allocated to locality pay.
Meanwhile, the FEHBlog wishes to point out that the omnibus includes the three now standard FEHB appropriations measures — the Hyde Amendment restrictions on abortion coverage (Division E summary at 63), the prohibition on applying full Cost Accounting Standards coverage to FEHB contracts (Division E summary, p. 93) and the contraceptive coverage mandate (Division E summary at 68).
What’s more, the OPM appropriations measures include the following
Exploring Tools for Prescription Drug Price Transparency in the Federal Employee Health Benefits (FEHB) Program.- OPM is directed to explore and evaluate the benefits and potential overall cost savings resulting from FEHB Carriers’ implementation of Internet-based self-service tools that deliver transparency and clinical decision support on prescription drug costs to its members. OPM is directed to report to the Committees one year after enactment of this Act, contingent on the availability of funding for this study.
In No Surprises Act news, the Internal Revenue Service issued guidance on calculating the qualifying payment amounts in 2023.
For qualifying payment amounts calculated by increasing the median contracted rate for 201913, the qualifying payment amounts for items and services furnished in 2023 are determined by taking the qualifying payment amounts calculated for items and services furnished in 2022 and multiplying the 2022 adjusted qualifying payment amounts by the percentage increase from 2022 to 2023, that is, 1.0768582128.
For example: An item is furnished in 2023. The median contracted rate for the item on January 31, 2019 was $1,500. The 2022 adjusted qualifying payment amount for the item was $1,597 ($1,500 x 1.0648523983). The 2023 adjusted qualifying payment amount for the item is $1,720 ($1,597 x 1.0768582128).
The notice also provides QPA adjustment guidance for plans that began after January 31, 2019.
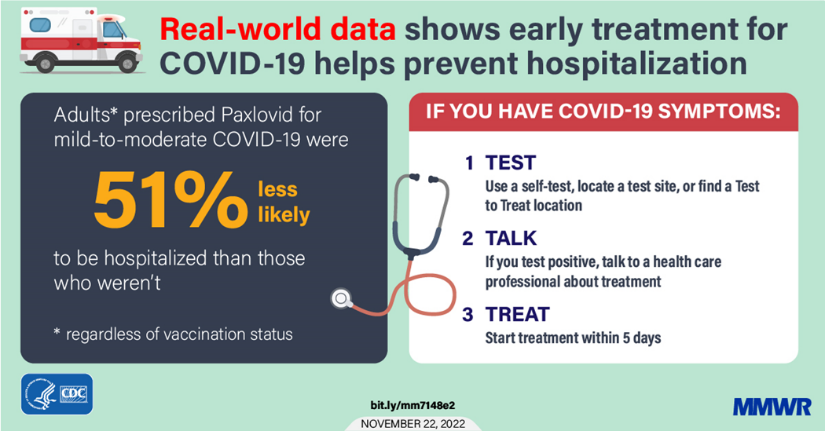
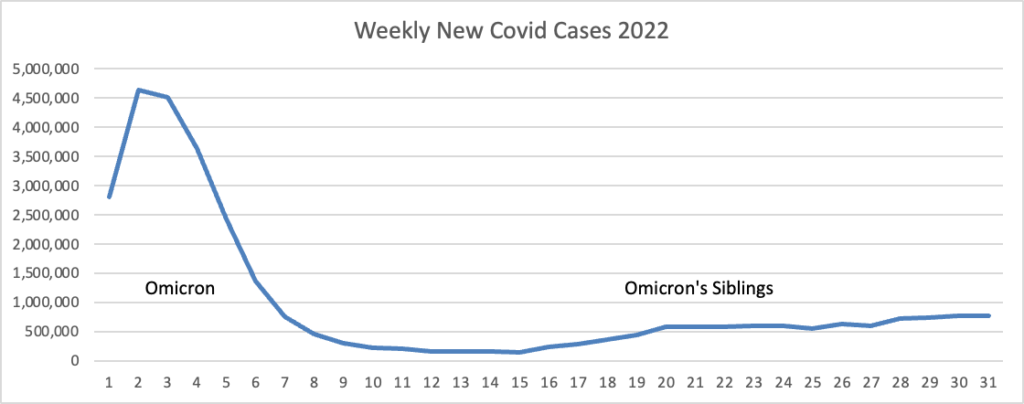
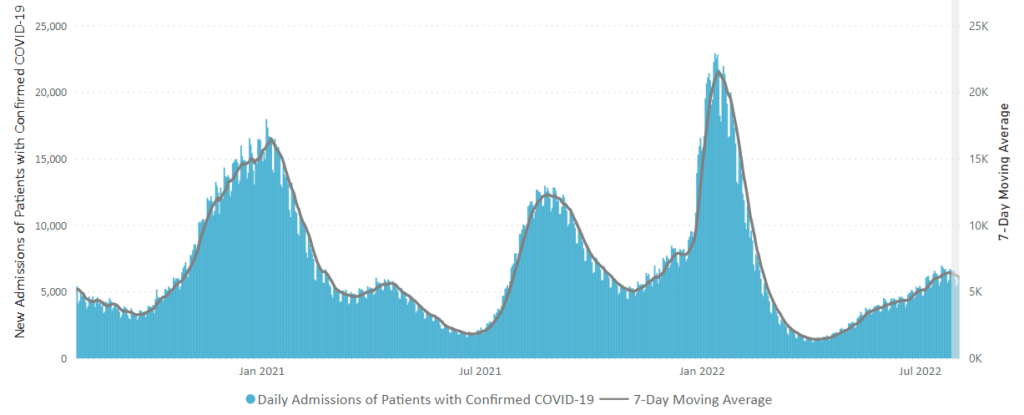
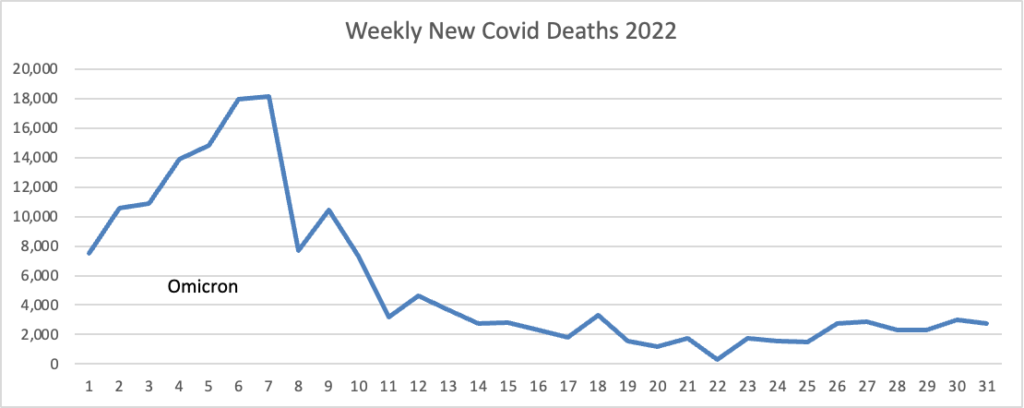
From the Omicron and siblings front, the Institute for Clinical and Economic Review (ICER) released “an update to the health benefit price benchmark for nirmatrelvir/ritonavir (Paxlovid™, Pfizer) for the treatment of COVID-19.”
Based on the current evidence, ICER’s health-benefit price benchmark (HBPB) for Paxlovid is $563-$906 per treatment course.
ICER’s HBPB is a price range suggesting the highest US price a manufacturer should charge for a treatment, based on the amount of improvement in overall health patients receive from that treatment, when a higher price would cause disproportionately greater losses in health among other patients in the health system due to rising overall costs of health care and health insurance. In short, it is the top price range at which a health system can reward innovation and better health for patients without doing more harm than good.
Of course, at this time, the federal government is covering the cost of Paxlovid for Americans. That may change in 2023 because, contrary to the FEHBlog’s expectation, the omnibus does not appear to include additional funding for Covid vaccines and treatment. However, the FEHBlog is confident that the federal government will find the money if it wants.
From the U.S. healthcare business front, Healthcare Dive tells us
- Looking to further boost its growing cell therapy business, Gilead Sciences on Wednesday said it plans to acquire Tmunity Therapeutics, a private biotechnology company trying to develop newer, better CAR-T treatments.
- CAR-T uses genetically engineered T cells to help the body fight diseases like cancer. Gilead currently markets two such products, Yescarta and Tecartus, which it obtained through the $12 billion purchase of Kite Pharma in 2017. Combined, sales of Yescarta and Tecartus were just under $400 million in the third quarter, a nearly 80% increase from the same three-month period a year prior.
- Gilead said that buying Tmunity should complement Kite’s cell therapy research capabilities by providing a new technology platform, a slate of preclinical- and clinical-stage programs, and a strategic partnership with the University of Pennsylvania. Financial terms of the acquisition weren’t disclosed. The companies expect their deal to close early next year.
In good news, Health Payer Intelligence informs us
The majority of Americans are satisfied with their employer-sponsored health insurance and cited it as the most important benefit an employer can offer, according to a poll conducted by Seven Letter Insight for the Protecting Americans’ Coverage Together (PACT) campaign.
The poll surveyed 2,334 individuals with employer-sponsored health plans between November 14 and November 19, 2022. * * *
Overall satisfaction with employer-sponsored coverage was also high. Most respondents (93 percent) said they were satisfied with their insurance, with 54 percent saying they were highly satisfied. Eighty-seven percent agreed that their plans were affordable, and 73 percent thought their insurance was worth what they paid.
When respondents were asked to describe their employer-sponsored coverage, affordable, high-quality, and comprehensive were the top descriptions, the survey noted.
From the miscellany department —
- Medscape provides an in-depth look at the progress in the fight against aging.
- Beckers Hospital Review identifies the top five patient safety issues for 2023.
- Govexec reports on the progress the federal government’s Merit Systems Performance Board has made since Congress restored the Board’s quorum last May after five years without one.