Happy Washington’s Birthday

The National Archives informs us
George Washington was born in Virginia on February 11, 1731, according to the then-used Julian calendar. In 1752, however, Britain and all its colonies adopted the Gregorian calendar which moved Washington’s birthday a year and 11 days to February 22, 1732.
Americans celebrated Washington’s Birthday long before Congress declared it a federal holiday. The centennial of his birth prompted festivities nationally and Congress established a Joint Committee to arrange for the occasion.
Washington’s Birthday was celebrated [as a federal holiday] on February 22nd [from 1879] until well into the 20th Century. However, in 1968 Congress passed the Monday Holiday Law to “provide uniform annual observances of certain legal public holidays on Mondays.” By creating more 3-day weekends, Congress hoped to “bring substantial benefits to both the spiritual and economic life of the Nation.”
One of the provisions of this act changed the observance of Washington’s Birthday from February 22nd to the third Monday in February. Ironically, this guaranteed that the holiday would never be celebrated on Washington’s actual birthday, as the third Monday in February cannot fall any later than February 21.
Contrary to popular belief, neither Congress nor the President has ever stipulated that the name of the holiday observed as Washington’s Birthday be changed to “President’s Day.”
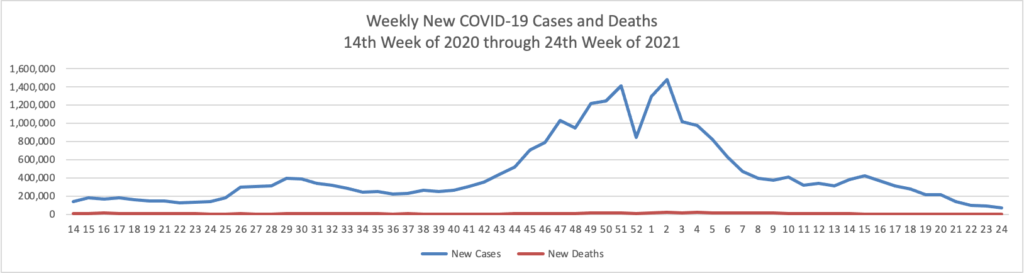
From the Omicron front, Medscape reports
Average daily COVID-19 cases and hospitalizations are continuing to fall in the U.S., providing a signal that the Omicron variant is receding across the country.
* * *
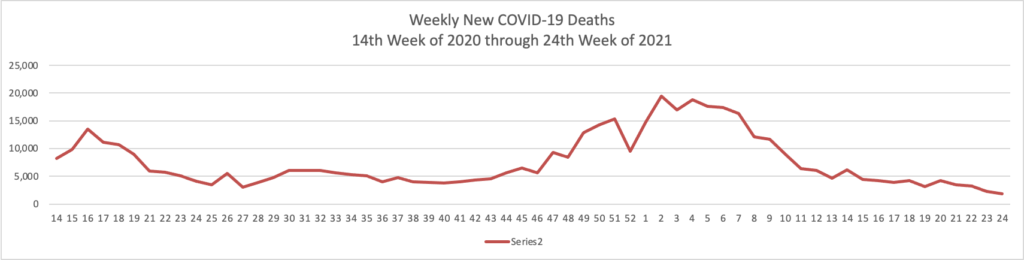
Deaths are also beginning to decline, with an average of about 2,100 daily, according to the data tracker from The New York Times. More than 2,500 daily deaths were being reported in early February.
Medpage Today adds
The Omicron sublineage BA.2, which raised alarms because of its transmission advantage, accounts for just 4% of cases in the U.S., and experts suggested it’s not likely to reverse the current downward trend of cases overall.
BA.2 has certainly increased in prevalence, but its been a slow trajectory, according to CDC data. BA.1.1, another Omicron sublineage, actually became the dominant strain in the U.S. in mid-January, the agency reported, with the original lineage, B.1.1.529, currently accounting for about 23% of U.S. cases.
“The BA.2 Omicron variant is increasing in prevalence slowly in both CDC and private data,” tweeted Scott Gottlieb, MD, former FDA commissioner and current Pfizer board member. “While it may become a dominant strain in time, it appears increasingly unlikely that it will cause a significant change in the downward trajectory of the current epidemic wave.”
That has certainly been the case for South Africa, where BA.2 accounts for nearly 100% of cases, according to Tulio de Oliveira, PhD, of the Centre for Epidemic Response & Innovation in Stellenbosch.
“This comes on a background of decreasing infections,” de Oliveira tweeted. The country now has a 7-day average of about 2,500 daily infections, down from a peak of about 23,000 in mid-December.
“At present, the Omicron BA.2 is not of great concern in South Africa,” he tweeted. “But our network is following very close and is alerted to its emergence.”
From the Covid vaccination mandate front, the Society for Human Resource Management reports that the U.S. Court of Appeals for the Fifth Circuit ordered the lower court to reconsider whether United Airlines vaccination mandate offered sufficient protection under Tittle VII to religious objectors.
From the No Surprises Act front, the American Hospital Association tells us
The Centers for Medicare & Medicaid Services will host a conference call for health care providers Feb. 23 at 2 p.m. ET on the No Surprises Act’s continuity of care, provider directory and public disclosure requirements. To participate in the Special Open Door Forum, dial 888-455-1397 and reference passcode 5109694. Slides for the call are available here. Participants may email questions in advance to Provider_Enforcement@cms.hhs.gov. A recording will be available after the call through Feb. 25 by dialing 866-373-4993.
The slide deck is quite detailed.
Healthcare Dive adds
The cost of being transported by ground ambulance has increased steadily over the past five years, according to a new report from nonprofit Fair Health, threatening patients with few protections from balance billing in disputes between insurers and ambulance providers.
More ambulance trips are billing payers for advanced life support ( ALS), denoting a higher level of care (and reimbursement) than basic life support (BLS) services. Private insurers’ average payments for those trips increased 56% between 2017 and 2020, from $486 to $758, according to the analysis. Before accounting for discounts negotiated with payers, the rate that ambulance operators charged for trips jumped 23% in the same time period and now average almost $1,300.
However, the average reimbursement for advanced life support ambulance rides climbed by 5% for patients covered by Medicare, from $441 to $463, suggesting the government program is keeping a lid on rising costs.
Although at this point only air ambulance charges are subject to the No Surprises Act, this type of report could lead Congress to expand the law to include ground ambulance charges.
From the medical research front, Biopharma Dive informs us
Eli Lilly is again upping its investment in genetic medicine, announcing Tuesday plans to establish a Boston research center that will use RNA- and DNA-based technologies to develop new drugs.
The Lilly Institute for Genetic Medicine will take up 334,000 square feet in Boston’s Seaport neighborhood, with occupancy slated to begin in 2024. Lilly, which has earmarked around $700 million for the project, expects the institute to grow from 120 employees to 250 within five years. Roles will include data scientists, chemists and research biologists with expertise in genetic medicine.
Alongside drug development, the institute will take a page from another Lilly venture — Lilly Gateway Labs — and offer shared lab and office space to support biotechnology startups in the area. Use of the shared space, Lilly said, could create as many as 150 additional new jobs once it’s fully occupied.
The National Institutes of Health announced
Women who had ever experienced sexual violence in their lifetime — including sexual assault and workplace sexual harassment — were more likely to develop high blood pressure over a seven-year follow-up period, according to findings from a large, longitudinal study of women in the United States. The research, funded by the National Institutes of Health and published in the Journal of the American Heart Association, indicated that sexual violence was a common experience, affecting more than 20% of the women in the sample.
“Our results showed that women who reported experiencing both sexual assault and workplace sexual harassment had the highest risk of hypertension, suggesting potential compounding effects of multiple sexual violence exposures on women’s cardiovascular health,” said Rebecca B. Lawn, Ph.D.(link is external), of the Harvard T. H. Chan School of Public Health, Boston, lead author on the study.
From the healthcare initiative front, the Centers for Disease Control detailed its Million Hearts 2027 initiative.
From the virtual care front, Healthcare IT News tells us
The American Medical Association, in conjunction with Manatt Health, published a report this week exploring the ways that virtual care and other digital tools can accelerate the adoption of the integrated delivery of behavioral and physical healthcare.
In the report, the organizations note that behavioral health integration is essential for solving the country’s dire need for access to services.
“The demand for behavioral health services is significant and rising, but so is the potential for digital technology to support the integrated delivery of physical and behavioral health services,” said AMA President Dr. Gerald Harmon in a statement.
Moreover, in Health Affairs, the National Coordinator for Health Information Technology Micky Tripathi discusses delivering on the promise of health information technology in 2022.
From the healthcare business front, Healthcare Finance reports
The Department of Justice has until Saturday, February 27 to block the merger between UnitedHealth Group and Change Healthcare, according to a Securities and Exchange Commission filing on February 17.
The filing, by Change, was done in accordance with a timing agreement made with the DOJ. In November 2021, the two companies agreed not to consummate their merger before February 22.
The timing agreement provides the DOJ with 10 days’ notice to sue to block the deal.
“Effective February 17, 2022, UnitedHealth Group and the Company provided such notice to the DOJ,” Change said in the filing. “Accordingly, the DOJ now has until February 27, 2022 to initiate litigation to block the consummation of the merger.”
The DOJ is preparing the lawsuit to block UnitedHealth from purchasing Change, according to Seeking Alpha.
From the awards front
- STAT News released its inaugural STATUS List which “recongizes standout individuals in health, medicine, and science. And although there are countless contenders to choose from, we’ve selected just 46 — an homage to the number of chromosomes in human DNA.”
- OPM “announced a call for nominations for the 2022 Presidential Rank Awards (PRAs). A Presidential Rank Award is one of the most prestigious awards in the federal career civil service, and these awards are critical to recognizing the hard work and important contributions of dedicated civil servants in the American federal workforce. The awards – which recognize exceptional performance by members of the career Senior Executive Service (SES) and Senior Career Employees – reflect the Biden-Harris Administration’s commitment to supporting the federal workforce and recognizing federal employees who serve with distinction.” The nomination deadline is March 25, 2022.