Friday Stats and More
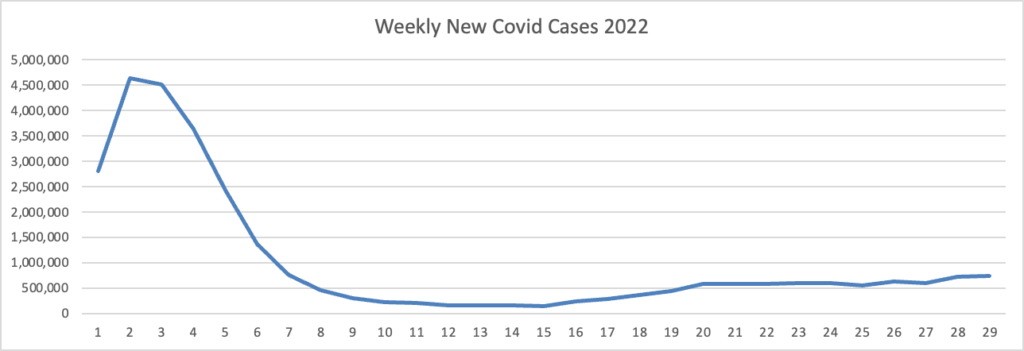
Based on the Centers for Disease Controls Covid Data Tracker and using Thursday as the first day of the week, here is the FEHBlog’s weekly chart of new Covid cases for 2022:
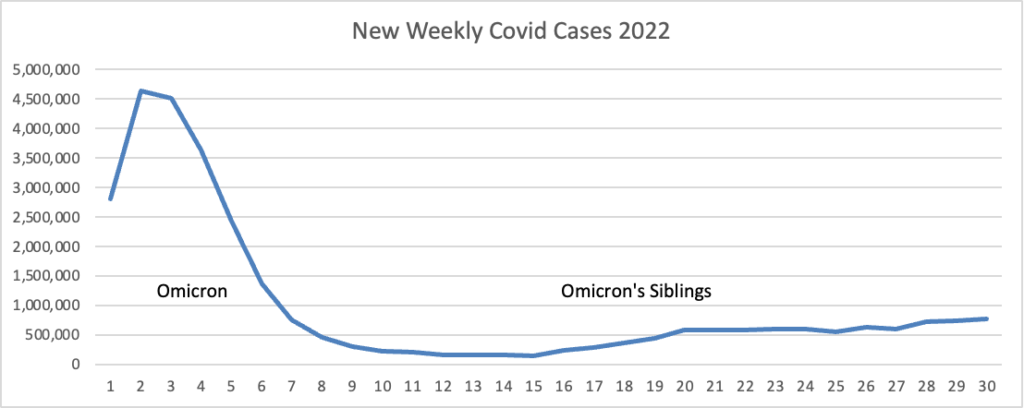
The CDC’s weekly review of its Covid statistics notes “As of July 27, 2022, the current 7-day moving average of daily new cases (126,272) decreased 0.9% compared with the previous 7-day moving average (127,478).”
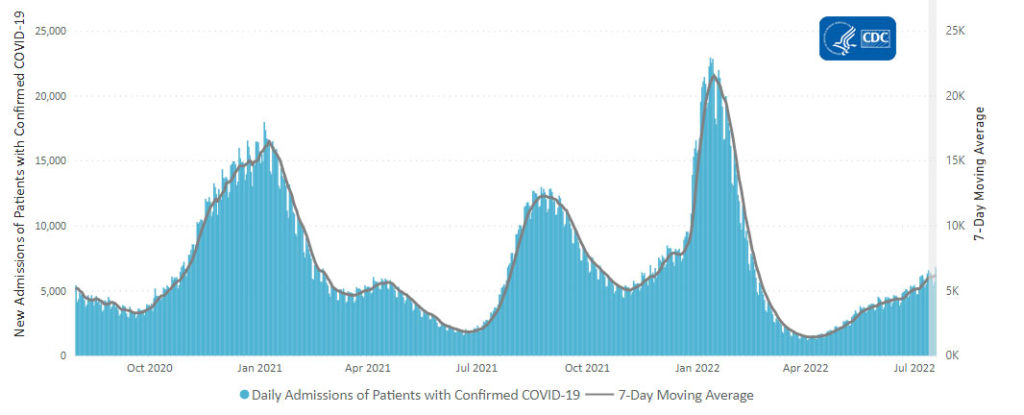
Here is the CDC’s chart of daily trends in new Covid hospital admissions:
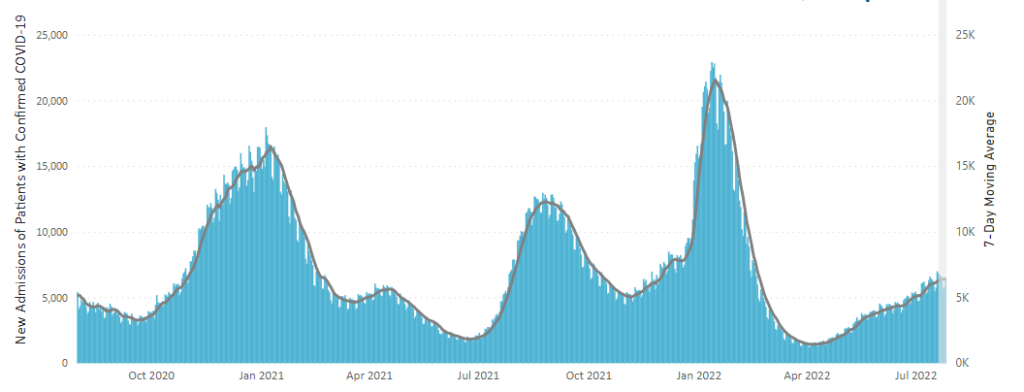
The CDC’s weekly review notes “The current 7-day daily average for July 20–26, 2022, was 6,340. This is a 1.7% increase from the prior 7-day average (6,231) from July 13–19, 2022.
Also, “CDC Nowcast projections for the week ending July 23, 2022, estimate that the combined national proportion of lineages designated as Omicron will continue to be 100% with the predominant Omicron lineage being BA.5, projected at 81.9% (95% PI 79.9-83.8%).”
STAT News reports
[New Yorker Janet] Handal and the roughly 200,000 other Americans to receive Evusheld have had to recalibrate their hopes. Although the Food and Drug Administration doubled the recommended dose to compensate, and the drug — when given along with the full complement of vaccines — still offers protection against severe disease, gone are hopes that the most vulnerable could ditch masks or return to their pre-Covid lives.
Although the problem is now eight months old, few other prophylactic drugs for people with compromised immune systems are on the horizon. Several companies have announced plans to bring forward new Covid-19 antibodies, but they virtually are all either still in animal studies or are being principally developed as therapeutics.
The absence is particularly notable given the potential size of the market: Although Evusheld has remained vastly underused, a fact some physicians attribute to its hazily defined efficacy, between 7 and 10 million Americans are immunocompromised — a huge market by drug industry standards.
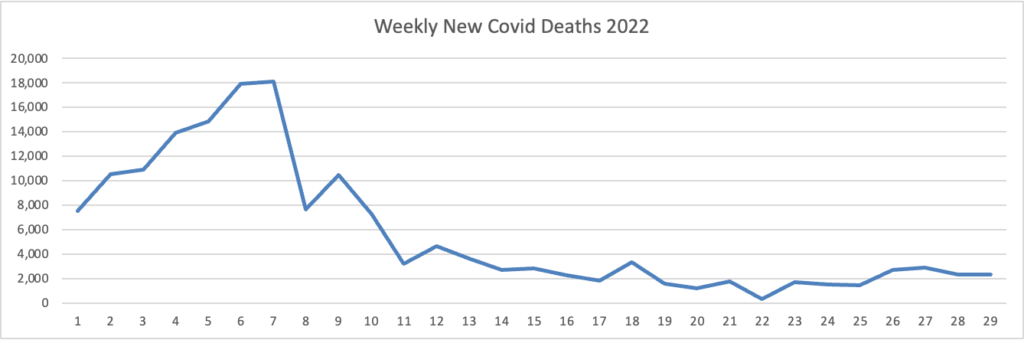
Here’s the FEHBlog’s weekly chart of new Covid deaths for 2022:
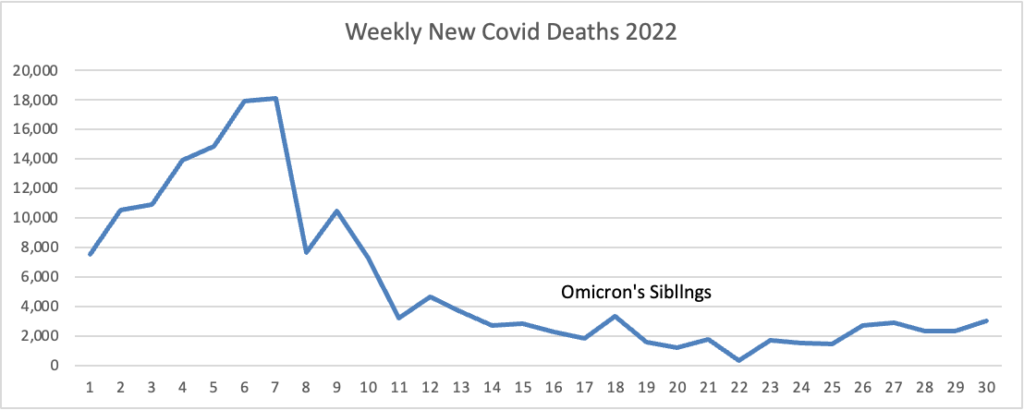
The CDC’s weekly review notes “The current 7-day moving average of new deaths (364) has decreased 4.8% compared with the previous 7-day moving average (382).”
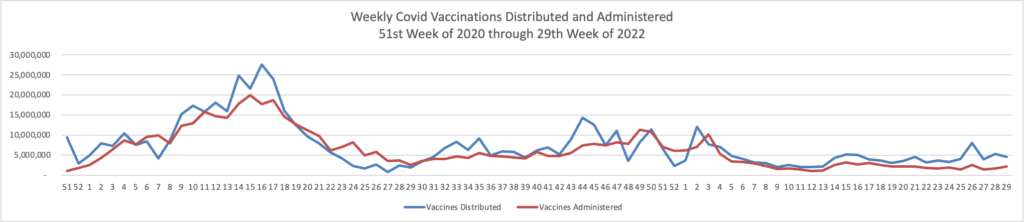
Here’s the FEHBlog’s weekly chart of Covid vaccinations distributed and administered from the 50th week of 2020, when the Covid vaccination era began to the 30th week of 2022:
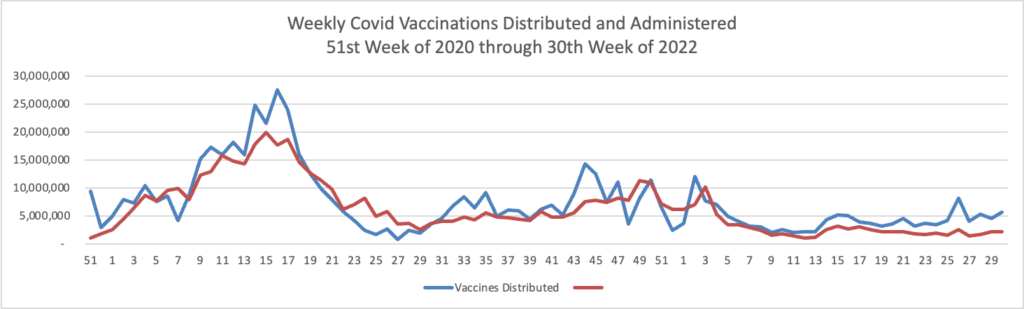
The CDC’s weekly review notes, “About 223.2 million people, or 67.2% of the total U.S. population, have been fully vaccinated.* Of those fully vaccinated, about 107.9 million people have received a booster dose, but 50.1% of the total booster-eligible population has not yet received a booster dose.”
Reuters reports
The U.S. government said on Friday it agreed to a $1.74 billion deal for 66 million doses of a Moderna COVID vaccine updated for the Omicron subvariant for use in a fall and winter booster campaign ***. Combined with 105 million doses the government already agreed to buy from Pfizer Inc and partner BioNTech SE for $3.2 billion, the latest deal puts the U.S. booster dose supply at about 171 million shots. read more
In CDC Communities News
As of July 28, 2022, there are 1,474 (45.8%) counties, districts, or territories with a high COVID-19 Community Level, 1,144 (35.5%) counties with a medium Community Level, and 602 (18.7%) counties with a low Community Level. Compared with last week, this represents a moderate increase (+3.9 percentage points) in the number of high-level counties, and a small decrease (−2.11 percentage points) in the number of medium-level counties and also a small decrease (−1.74 percentage points) in the number of low-level counties. 49 out of 52 jurisdictions* had high- or medium-level counties this week. Rhode Island, New Hampshire, and the District of Columbia are the only jurisdictions to have all counties at low Community Levels.
To check your COVID-19 Community Level, visit COVID Data Tracker. To learn which prevention measures are recommended based on your COVID-19 Community Level, visit COVID-19 Community Level and COVID-19 Prevention.
From the monkeypox front, the FDA provides an update on the agency’s response to this outbreak.
From the mental healthcare front —
MedPage Today brings us encouraging news
Local outpatient mental health providers are doing fine handling the increased demand from the newly launched nationwide “988” Suicide and Crisis Lifeline, despite fears that they wouldn’t be ready, according to a senior administration official.
“The states and call centers across the country absolutely rose to the increased volume that we saw,” the official said Thursday evening during a background briefing on distribution of federal grants to shore up mental health services in schools. “We can see a 45% increase in the volume of calls that came in the week of the launch, compared to the week prior, across the country — an additional 23,000 … calls, texts, and chats that came in across the lifeline.”
Healthcare Dive reports that CVS Health has appointed its first chief psychiatric officer. “Taft Parsons, MD, who has previously worked for Molina Healthcare and Henry Ford Health System, will be tasked with focusing on mental well-being for patients and will lead the Aetna Medical Affairs Behavioral Health organization.” Good luck, Dr. Parsons.
From the medical research front —
- HealthDay informs us “Obesity, depression, high blood pressure, asthma: These are just a few of the chronic health conditions that are now affecting almost 40 million Americans between the ages 18 and 34, new federal data shows.”
- Mhealth Intelligence adds “While discovering new methods of care during the COVID-19 pandemic, Stanford Medicine researchers found that a smartphone app known as SkinIO is highly resourceful when detecting skin cancer among older people.”
From the telehealth front —
- UPI reports “Using telehealth to supplement or replace in-person maternal care services, such as postpartum depression and diabetes and hypertension monitoring during pregnancy, leads to similar — and sometimes better — clinical outcomes and patient satisfaction compared to in-person care. That’s the gist of a study whose findings were published Monday in Annals of Internal Medicine.”
- The Department of Health and Human Services and the Justice Department released guidance that they are partnering to enforce various federal civil rights laws “requiring that telehealth be accessible to people with disabilities and limited English proficient persons. These laws work in tandem to prohibit discrimination and protect access to health care. The guidance may be found at: https://www.hhs.gov/sites/default/files/guidance-on-nondiscrimination-in-telehealth.pdf – PDF.”
Finally, Health Payer Intelligence discusses a strategy for communicating relating high health plan premium increases for next year.
There are two key factors that will influence employer-sponsored health plan premiums in 2023, Jim Winkler, chief strategy officer at the Business Group on Health, told HealthPayerIntelligence by email:
* Market inflation on prices of healthcare services
* Utilization rate of healthcare services
“Heading into 2023, employers face uncertainty in terms of the impact of market inflation on the prices of health care services and potential increases in utilization of those services,” Winkler said.
“This all comes at a time when employers also have a heightened focus on attracting and retaining key talent while addressing the overall health and well-being needs of the workforce and their families.” * * *
“As employers prepare to communicate with their workforce, they will see that employees tend to be focused on what any cost increase means to them, both in terms of out-of-pocket costs and paycheck deductions. Employers will therefore need to assist employees on understanding how best to manage their own costs,” Winkler recommended.
Omnichannel communication with health plan members can be particularly useful for improving member engagement in employer-sponsored health plans. With omnichannel communication, payers and employers coordinate communication across multiple channels and platforms, improving the timeliness and convenience of member interactions.